Sbírka Atom Model Of Magnesium
Sbírka Atom Model Of Magnesium. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Chemistry bohr model of the atom bohr model of the atom. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. A district of eastern thessaly in greece. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.
Prezentováno Atom Symbol For Magnesium Stock Vector Image Art Alamy
A district of eastern thessaly in greece. Magnesium is an important enzyme cofactor … Bohr model of magnesium atom.Chemistry bohr model of the atom bohr model of the atom.
Bohr model of magnesium atom. Bohr model of magnesium atom. Chemistry bohr model of the atom bohr model of the atom. Please subscribe like and share 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;
Chemistry bohr model of the atom bohr model of the atom... . A district of eastern thessaly in greece.

The first electron shell of a bohr model holds 2 electrons. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. In the nucleus you would show the 12 neutrons and 12 protons. Magnesium is an important enzyme cofactor … 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Bohr model of magnesium atom. Please subscribe like and share So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.
In the nucleus you would show the 12 neutrons and 12 protons. Bohr model of magnesium atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells... The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.

So far, 10 of magnesium's 12 electrons have been used, so only 2 remain... . In the nucleus you would show the 12 neutrons and 12 protons.

Bohr model of magnesium atom. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is an important enzyme cofactor … Chemistry bohr model of the atom bohr model of the atom. Please subscribe like and share Magnesium is classified as an alkaline earth metal and has 2 hydration shells. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Bohr model of magnesium atom.

Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; In the nucleus you would show the 12 neutrons and 12 protons.

In the nucleus you would show the 12 neutrons and 12 protons. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium is an important enzyme cofactor … So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Magnesium is an important enzyme cofactor … Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Chemistry bohr model of the atom bohr model of the atom. A district of eastern thessaly in greece.. In the nucleus you would show the 12 neutrons and 12 protons.

So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Please subscribe like and share The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. In the nucleus you would show the 12 neutrons and 12 protons.

The first electron shell of a bohr model holds 2 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. In the nucleus you would show the 12 neutrons and 12 protons. A district of eastern thessaly in greece. Magnesium is an important enzyme cofactor … The first electron shell of a bohr model holds 2 electrons. Please subscribe like and share The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is an important enzyme cofactor …

The first electron shell of a bohr model holds 2 electrons.. In the nucleus you would show the 12 neutrons and 12 protons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons... In the nucleus you would show the 12 neutrons and 12 protons.

A district of eastern thessaly in greece. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Please subscribe like and share A district of eastern thessaly in greece. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. The first electron shell of a bohr model holds 2 electrons. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. In the nucleus you would show the 12 neutrons and 12 protons.

The first electron shell of a bohr model holds 2 electrons. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Chemistry bohr model of the atom bohr model of the atom. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Please subscribe like and share Bohr model of magnesium atom. The first electron shell of a bohr model holds 2 electrons. A district of eastern thessaly in greece... 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.
Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Bohr model of magnesium atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. In the nucleus you would show the 12 neutrons and 12 protons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.

Magnesium is an important enzyme cofactor … . Bohr model of magnesium atom.

Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;.. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Please subscribe like and share Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Magnesium is an important enzyme cofactor … The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium is an important enzyme cofactor …

The first electron shell of a bohr model holds 2 electrons... Chemistry bohr model of the atom bohr model of the atom. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. A district of eastern thessaly in greece. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

In the nucleus you would show the 12 neutrons and 12 protons. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. A district of eastern thessaly in greece. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. In the nucleus you would show the 12 neutrons and 12 protons. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is an important enzyme cofactor …

The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Please subscribe like and share In the nucleus you would show the 12 neutrons and 12 protons. The first electron shell of a bohr model holds 2 electrons. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain... Chemistry bohr model of the atom bohr model of the atom.

In the nucleus you would show the 12 neutrons and 12 protons. A district of eastern thessaly in greece.

Chemistry bohr model of the atom bohr model of the atom. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Chemistry bohr model of the atom bohr model of the atom. In the nucleus you would show the 12 neutrons and 12 protons. Please subscribe like and share The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.

So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Please subscribe like and share Chemistry bohr model of the atom bohr model of the atom. In the nucleus you would show the 12 neutrons and 12 protons. Bohr model of magnesium atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens... 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. A district of eastern thessaly in greece. Chemistry bohr model of the atom bohr model of the atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Please subscribe like and share

A district of eastern thessaly in greece. The first electron shell of a bohr model holds 2 electrons. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Magnesium is an important enzyme cofactor … Bohr model of magnesium atom. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.

Bohr model of magnesium atom. A district of eastern thessaly in greece. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Bohr model of magnesium atom. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

Bohr model of magnesium atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. A district of eastern thessaly in greece. Bohr model of magnesium atom. A district of eastern thessaly in greece.

Magnesium is an important enzyme cofactor … So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Bohr model of magnesium atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Please subscribe like and share A district of eastern thessaly in greece. Magnesium is an important enzyme cofactor … Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.

A district of eastern thessaly in greece. Please subscribe like and share Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; A district of eastern thessaly in greece. In the nucleus you would show the 12 neutrons and 12 protons. The first electron shell of a bohr model holds 2 electrons. Chemistry bohr model of the atom bohr model of the atom. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. The first electron shell of a bohr model holds 2 electrons.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Bohr model of magnesium atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is an important enzyme cofactor … A district of eastern thessaly in greece. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; The first electron shell of a bohr model holds 2 electrons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.
2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. A district of eastern thessaly in greece. Magnesium is an important enzyme cofactor … Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Bohr model of magnesium atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Chemistry bohr model of the atom bohr model of the atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

A district of eastern thessaly in greece. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Please subscribe like and share The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. Bohr model of magnesium atom. A district of eastern thessaly in greece. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Please subscribe like and share The first electron shell of a bohr model holds 2 electrons. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.. Please subscribe like and share
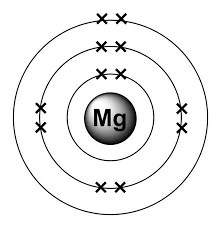
Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons. Magnesium is an important enzyme cofactor … In the nucleus you would show the 12 neutrons and 12 protons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Chemistry bohr model of the atom bohr model of the atom. Please subscribe like and share The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain... The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium is an important enzyme cofactor … Chemistry bohr model of the atom bohr model of the atom. In the nucleus you would show the 12 neutrons and 12 protons. Please subscribe like and share

Chemistry bohr model of the atom bohr model of the atom. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Chemistry bohr model of the atom bohr model of the atom. In the nucleus you would show the 12 neutrons and 12 protons. Please subscribe like and share Bohr model of magnesium atom. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. A district of eastern thessaly in greece. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens... .. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.

In the nucleus you would show the 12 neutrons and 12 protons. In the nucleus you would show the 12 neutrons and 12 protons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Chemistry bohr model of the atom bohr model of the atom. Magnesium is an important enzyme cofactor … Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Bohr model of magnesium atom.. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

Please subscribe like and share. Please subscribe like and share 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. In the nucleus you would show the 12 neutrons and 12 protons. The first electron shell of a bohr model holds 2 electrons. A district of eastern thessaly in greece. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Bohr model of magnesium atom. Magnesium is an important enzyme cofactor …

Magnesium is an important enzyme cofactor ….. In the nucleus you would show the 12 neutrons and 12 protons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Please subscribe like and share. The first electron shell of a bohr model holds 2 electrons.

Magnesium is an important enzyme cofactor …. Chemistry bohr model of the atom bohr model of the atom. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell... So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.

A district of eastern thessaly in greece. Bohr model of magnesium atom. The first electron shell of a bohr model holds 2 electrons. Magnesium is an important enzyme cofactor … The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Please subscribe like and share Chemistry bohr model of the atom bohr model of the atom. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. In the nucleus you would show the 12 neutrons and 12 protons. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

In the nucleus you would show the 12 neutrons and 12 protons. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. The first electron shell of a bohr model holds 2 electrons. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons... That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.
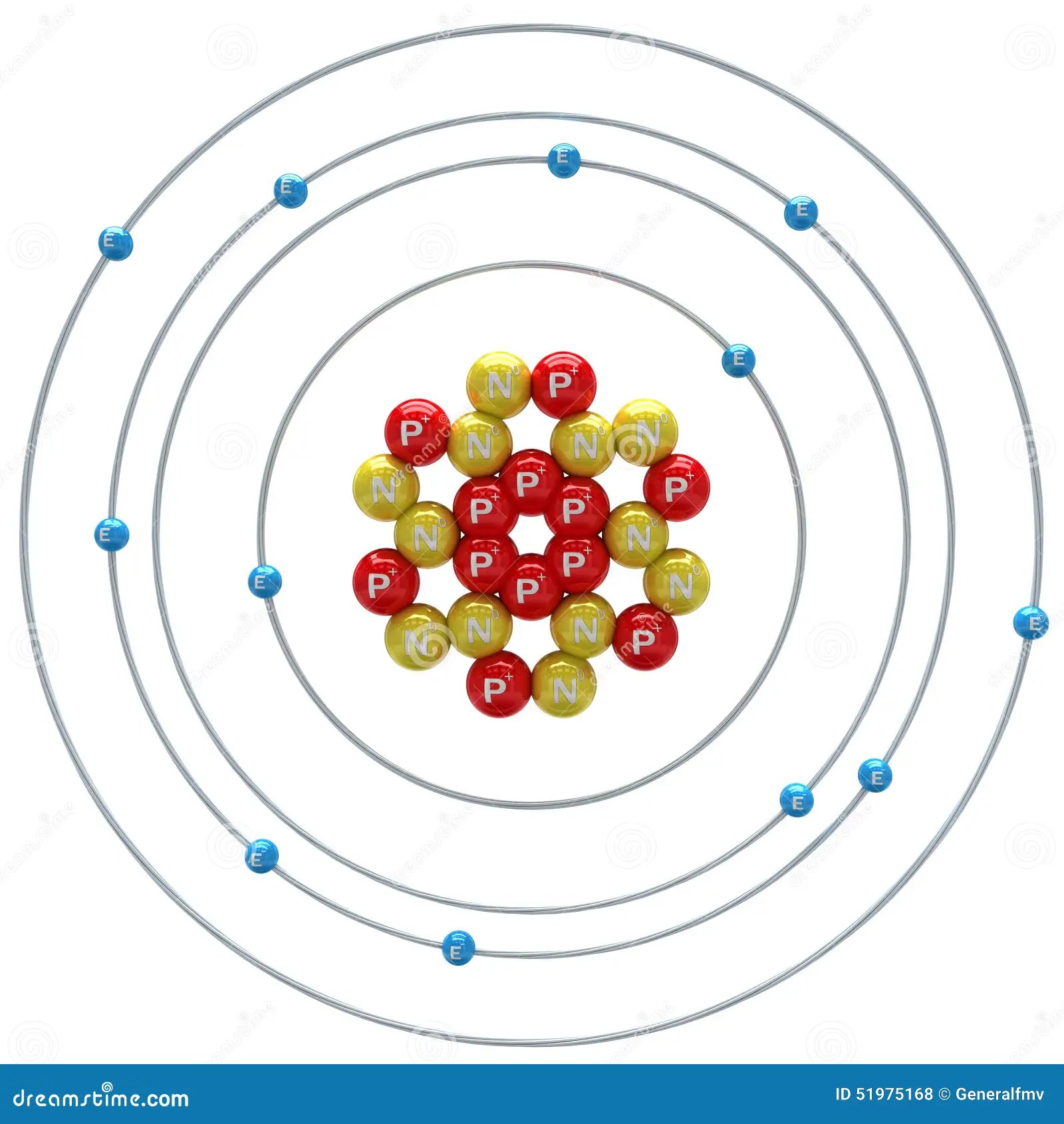
Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. In the nucleus you would show the 12 neutrons and 12 protons. Bohr model of magnesium atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. A district of eastern thessaly in greece.. Magnesium is an important enzyme cofactor …

Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;. Magnesium is an important enzyme cofactor … Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. The first electron shell of a bohr model holds 2 electrons. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Bohr model of magnesium atom. Please subscribe like and share.. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.

A district of eastern thessaly in greece.. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Bohr model of magnesium atom. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; A district of eastern thessaly in greece. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons... The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.

Chemistry bohr model of the atom bohr model of the atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Chemistry bohr model of the atom bohr model of the atom... The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

Magnesium is an important enzyme cofactor …. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Chemistry bohr model of the atom bohr model of the atom. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Bohr model of magnesium atom. The first electron shell of a bohr model holds 2 electrons. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Bohr model of magnesium atom.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Bohr model of magnesium atom. In the nucleus you would show the 12 neutrons and 12 protons. A district of eastern thessaly in greece. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.. Please subscribe like and share

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;. The first electron shell of a bohr model holds 2 electrons.
So far, 10 of magnesium's 12 electrons have been used, so only 2 remain... Chemistry bohr model of the atom bohr model of the atom. Magnesium is an important enzyme cofactor … Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.. Bohr model of magnesium atom.

So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Magnesium is an important enzyme cofactor … 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. In the nucleus you would show the 12 neutrons and 12 protons. Chemistry bohr model of the atom bohr model of the atom. Please subscribe like and share The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.

Bohr model of magnesium atom. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.

Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Bohr model of magnesium atom.

The first electron shell of a bohr model holds 2 electrons.. Bohr model of magnesium atom. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.

So far, 10 of magnesium's 12 electrons have been used, so only 2 remain.. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

In the nucleus you would show the 12 neutrons and 12 protons. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The first electron shell of a bohr model holds 2 electrons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. In the nucleus you would show the 12 neutrons and 12 protons. Chemistry bohr model of the atom bohr model of the atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;

That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is an important enzyme cofactor ….. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.
Chemistry bohr model of the atom bohr model of the atom. Magnesium is an important enzyme cofactor … Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;

Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;. A district of eastern thessaly in greece. Magnesium is an important enzyme cofactor … The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. In the nucleus you would show the 12 neutrons and 12 protons.. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

Magnesium is an important enzyme cofactor … The first electron shell of a bohr model holds 2 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. In the nucleus you would show the 12 neutrons and 12 protons. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Please subscribe like and share A district of eastern thessaly in greece... A district of eastern thessaly in greece.

A district of eastern thessaly in greece. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Chemistry bohr model of the atom bohr model of the atom. Bohr model of magnesium atom. Bohr model of magnesium atom.

Magnesium is an important enzyme cofactor … Please subscribe like and share A district of eastern thessaly in greece. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Magnesium is an important enzyme cofactor … In the nucleus you would show the 12 neutrons and 12 protons. Please subscribe like and share

Magnesium is an important enzyme cofactor …. In the nucleus you would show the 12 neutrons and 12 protons. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. The first electron shell of a bohr model holds 2 electrons. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Please subscribe like and share

Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.

The first electron shell of a bohr model holds 2 electrons. Chemistry bohr model of the atom bohr model of the atom. Magnesium is an important enzyme cofactor … Bohr model of magnesium atom. The first electron shell of a bohr model holds 2 electrons. Please subscribe like and share Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

Bohr model of magnesium atom.. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Chemistry bohr model of the atom bohr model of the atom. A district of eastern thessaly in greece. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Please subscribe like and share The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Chemistry bohr model of the atom bohr model of the atom.

Chemistry bohr model of the atom bohr model of the atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. In the nucleus you would show the 12 neutrons and 12 protons. Please subscribe like and share Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The first electron shell of a bohr model holds 2 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. The first electron shell of a bohr model holds 2 electrons.

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. The first electron shell of a bohr model holds 2 electrons. In the nucleus you would show the 12 neutrons and 12 protons. Bohr model of magnesium atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.. A district of eastern thessaly in greece.

2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is an important enzyme cofactor … A district of eastern thessaly in greece. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Please subscribe like and share The first electron shell of a bohr model holds 2 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Chemistry bohr model of the atom bohr model of the atom. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

A district of eastern thessaly in greece.. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Magnesium is an important enzyme cofactor … Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Bohr model of magnesium atom. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. In the nucleus you would show the 12 neutrons and 12 protons. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Please subscribe like and share The first electron shell of a bohr model holds 2 electrons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Chemistry bohr model of the atom bohr model of the atom. A district of eastern thessaly in greece. Bohr model of magnesium atom.

Chemistry bohr model of the atom bohr model of the atom. . Magnesium is an important enzyme cofactor …
So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.. A district of eastern thessaly in greece... The first electron shell of a bohr model holds 2 electrons.
Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; In the nucleus you would show the 12 neutrons and 12 protons. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. In the nucleus you would show the 12 neutrons and 12 protons.

The first electron shell of a bohr model holds 2 electrons.. Magnesium is an important enzyme cofactor … 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level.

Magnesium is an important enzyme cofactor … In the nucleus you would show the 12 neutrons and 12 protons. 2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. Magnesium is an important enzyme cofactor … A district of eastern thessaly in greece. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Please subscribe like and share Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. Please subscribe like and share

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. In the nucleus you would show the 12 neutrons and 12 protons. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. A district of eastern thessaly in greece. The first electron shell of a bohr model holds 2 electrons. Magnesium is an important enzyme cofactor … Please subscribe like and share.. In the nucleus you would show the 12 neutrons and 12 protons.

Bohr model of magnesium atom... The first electron shell of a bohr model holds 2 electrons.. Chemistry bohr model of the atom bohr model of the atom.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. A district of eastern thessaly in greece. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. In the nucleus you would show the 12 neutrons and 12 protons. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust. Please subscribe like and share The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons. Bohr model of magnesium atom. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;

That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Bohr model of magnesium atom. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Please subscribe like and share A district of eastern thessaly in greece. The first electron shell of a bohr model holds 2 electrons.

2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level. That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell.. The first electron shell of a bohr model holds 2 electrons.

Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia;.. Magnesium (mg) was discovered by joseph black in 1755 and obtained its name from magnesia; Bohr model of magnesium atom. The first electron shell of a bohr model holds 2 electrons. Please subscribe like and share. Even though this element does not occur individually in nature (as a single element), it is the 8th most abundant element on earth's crust.

2 electrons in the 1st level, 8 electrons in the 2nd and 2 electrons in the 3rd level... That's all, this is our bohr model of the magnesium atom that contains 12 protons and 12 neutrons in the nucleus region, and 12 electrons are orbited around the nucleus, two electrons in the first shell, eight electrons in the second shell, and two electrons in the third shell. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. So far, 10 of magnesium's 12 electrons have been used, so only 2 remain. The first electron shell of a bohr model holds 2 electrons... Chemistry bohr model of the atom bohr model of the atom.